This paper was originally published at the Society of Plastics Engineers Annual Technical Conference 2013.
Abstract
There are multiple applications where chlorinated poly(vinyl chloride) (CPVC) may come in contact with glycerin. One common application is in fire suppression systems that could be subjected to subfreezing temperatures. Chlorinated poly(vinyl chloride) is increasingly being used for these systems in place of metal because of its many advantages, including the ease of installation, weight reduction, cost benefits and chemical resistance. When CPVC piping is used in an area that has the potential to freeze, an antifreeze solution must be used in the fire suppression systems to suppress the freezing temperature of the water and reduce possibility of failure of the piping system. Glycerin is a commonly used antifreeze for this application. The following article discusses the effects of using glycerin with CPVC piping and presents a case study of the use of bio-derived glycerin as an antifreeze agent. In general, it was found that glycerin from the bio-diesel industry had adverse effects on the CPVC.
Introduction
A common application where CPVC may come into contact with glycerin is in fire suppression systems. It has been proven that one of the most effective techniques to prevent a fire from spreading is the use of ceiling mounted fire sprinklers. Their use in new commercial construction has been required for many years. In addition, requirements of their use in multifamily and even single family residence has become increasing popular. A proper sprinkler layout requires the connection of numerous pipe sections routed throughout the building’s ceiling and walls.
Chlorinated poly(vinyl chloride) piping is increasingly being used in place of metal piping because of its many advantageous, including the ease of installation, weight reduction, cost benefits and chemical resistance. However, when used as part of a fire suppression system in an area that has the potential to freeze, an antifreeze solution must be used to ensure freezing water does not cause failure of the plastic piping. Water freezing and expanding in a piping system can create excessively high pressures that can lead to a pipe or component failure. Freeze can cause water leakage from a pipe rupture or make the system ineffective in the event of a fire, as well as cause water damage if left unattended.
Common antifreeze agents used for fire suppression systems include chemicals such as propylene glycol and glycerin. These chemicals are used as antifreeze agents because their chemical structure forms strong hydrogen bonds with water molecules that prevent the formation of ice crystals at normal water freeze temperature, which suppresses the freezing temperature of the water. These chemicals are preferred over other antifreeze agents such as ethylene glycol because of their low toxicity. However, certain solutions of these chemicals can negatively affect CPVC piping [1]. Additionally, the specific method to produce these chemicals can result in various residual components. For example, glycerin can be produced through different methods, i.e. from propylene, as a byproduct of making soap, or as a co-product of biodiesel. Each process can yield various residual components within the glycerin. It is important to consider all these individual components present in an antifreeze solution that may come into contact with CPVC piping to assure that the integrity of the piping will be maintained during the intended service life.
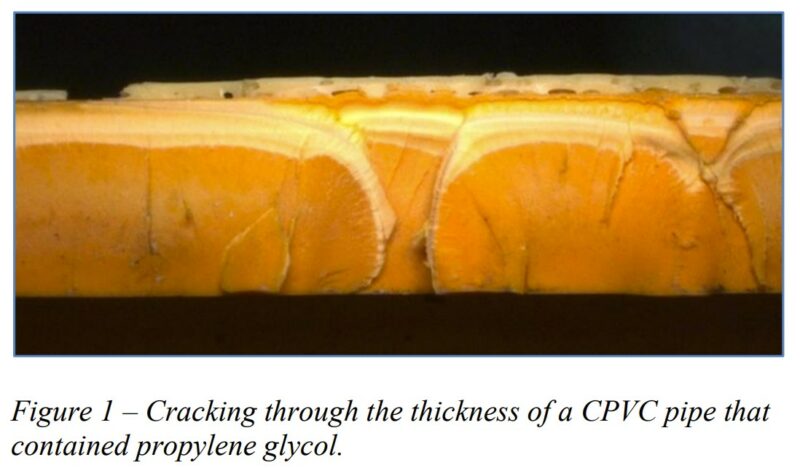
Contributors include: Paul J. Gramann, Ph.D., and Javier C. Cruz, Ph.D., The Madison Group; David Grewell, Ph.D, Melissa Montablo-Lomboy, Ph.D, Tong Wang, Ph.D., Iowa State University
