The adoption of plastic materials for outdoor applications has been an evolving process. During the early to mid-20th century, most plastics were often characterized as too brittle, color-fading and susceptible to rapid degradation when exposed to sunlight’s ultraviolet (UV) radiation. The first polymers to gain broader acceptance in outdoor applications were polyethylene (PE) and polyvinyl chloride (PVC) — materials that are still widely used today. In fact, PE and PVC are considered some of the most widely used plastics globally.

Cracks and faded colors are telltale signs of UV degradation in plastic parts that stay outside.
The use of PE in outdoor environments became more viable following the development of high-density polyethylene (HDPE). Prior to that, the polymer didn’t fare well with UV exposure. The development of stronger and more rigid HDPE coincided with significant progress in additive technologies for stabilization. These advances marked the beginning of a new era for polyolefins and many other polymers as durable outdoor materials.
PVC, first synthesized in the mid-19th century, was initially brittle and difficult to process. It was only after significant formulation advancements, particularly through the innovative incorporation of additives, that the material became both processable and more durable. After the 1950s, rigid PVC gained popularity for piping in distribution systems, although these installations typically involved limited to no direct exposure to sunlight.
I remember in the 1980s when plastic furniture was becoming more mainstream, having PVC plastic patio chairs next to our outdoor pool. Living close to the ocean, they were highly desirable as an alternative that would not corrode with the salty air. However, the chairs did not perform as well as expected and quickly chalked, eventually suffering brittle cracking with minimal force due to UV exposure. Formulations have improved immensely since then, and the incorporation of additives with synergistic property enhancements has substantially extended their durability. Today PVC is used in many outdoor products including commercial and residential construction materials like siding, windows and trim with lifespans exceeding 20 years of outdoor use.
These examples illustrate the broader evolution of plastics in outdoor applications. Today, there are many materials that perform extremely well when exposed to the sun’s intense UV rays. Identifying the ideal candidate for an outdoor application can be difficult with many materials marketed as being applicable for outdoor use. However, the inherent chemical structure of some polymers enables them to perform better than others.

Materials for Outdoor Applications
The resistance of a polymer to UV rays is directly related to its chemical structure and formulation. UV-induced degradation occurs when the polymer backbone absorbs UV radiation, disrupting its molecular structure. Generally, backbone rupture occurs from a combination of UV energy and the presence of an oxidizing agent such as atmospheric oxygen. The mechanism can be complex and result in cyclic reactions that lead to accelerated degradation (Figure 1). Materials that are sensitive to UV radiation are primarily those containing chromophores that absorb energy in the UV frequency range.
A chromophore is a location where the polymer can absorb wavelengths of light. While light absorption is critical to produce color, the absorption of shorter UV wavelengths can result in backbone rupture due to the higher energy at those frequencies. Chromophores are not always part of the standard polymer structure. Additional sources of chromophores may include a molecular defect within the polymer, another formulation constituent or a contaminant.
A good example of this is PE, which does not include chromophores within the backbone structure. However, non-stabilized PE is susceptible to UV degradation due to carbonyl and hydroperoxide groups introduced by structural anomalies. In fact, most polymers include contaminants and structural anomalies. As a result, even polymers typically considered UV stable often require stabilization additives to ensure adequate performance.
Three main factors influence a polymer’s ability to withstand UV exposure:
- The frequency of wavelengths it will absorb with shorter wavelengths being more damaging.
- The strength of the bonds of the backbone.
- The ability to dissipate the energy rather than absorb it.
Some polymers are inherently more UV resistant. Examples include:
Acrylics such as:
- Polymethyl methacrylate (PMMA)
- Acrylonitrile styrene acrylate (ASA)
Imides such as:
- Polyimide (PI)
- Polyether-imide (PEI)
- Polyamide-imide (PAI)
Fluoropolymers such as:
- Polytetrafluoroethylene (PTFE)
- Polyvinylidene fluoride (PVDF)
- Fluorinated ethylene propylene (FEP)
- Perfluoroalkoxy alkane (PFA)
These polymers, such as PMMA and ASA, are either mostly invisible to UV light or have extremely stable backbones that resist degradation (e.g., carbon-fluorine bonds in fluoropolymers). Many other polymers that aren’t naturally UV resistant can be made suitable for outdoor use through the use of various stabilization technologies. These additives or processes help polymers withstand UV exposure and maintain their performance. Here’s a breakdown of common polymers and their UV stability:
- Polycarbonate (PC) – Considered UV stable due to minimal mechanical property loss from UV attack, but subject to yellowing requiring blue-hue-correction additives.
- Polyphenylene sulfide (PPS) – Exhibits very good outdoor durability with proper stabilization.
- Polysulfone (PSU) – Unfilled versions are UV sensitive, but adding carbon black or other stabilizers improves their outdoor performance.
- Polypropylene (PP) and polyethylene (PE) – Require proper stabilization with HDPE being the most recommended and best formulated for outdoor applications.
- Polyphenylene oxide (PPO) and polybutylene terephthalate (PBT) – Both perform well outdoors when stabilized.
- Polyamide (PA 6, PA 6/6) – All grades need stabilization. For the most common grades, PA 6/6 is more sensitive to UV light than PA 6.
- Polyetherether ketone (PEEK) – While gamma-radiation resistant, it is UV sensitive and unsuitable for prolonged outdoor exposure.
- Polyoxymethylene (POM) & acrylonitrile butadiene styrene (ABS) – Although they can be stabilized, they are generally less suitable for long-term outdoor use.
Picking the Right Plastic
An effective material selection process begins with the early identification of all critical performance requirements. For an outdoor application, it’s essential to develop a comprehensive specification list, rather than focusing primarily on UV resistance. This broader approach increases the likelihood of success, as material options will be tailored to meet all critical property requirements and not just resistance to photo-oxidation. While UV degradation is an obvious consideration, there are many other factors that need to be considered that could have a greater effect on the final product performance. Critical properties to consider include:
- Mechanical strength
- Impact resistance
- Thermal transitions
- Coefficient of thermal expansion
- Ductile-to-brittle transitions
- Temperature (oxidation) resistance
- Creep and fatigue
- Friction and abrasion
- Density and weight
- Cost
- Recyclability
In addition, multiple outdoor environmental factors beyond UV resistance need to be considered including:
- Environmental impact
- Humidity and moisture effects from rain and dew (including hydrolysis resistance and aesthetics)
- Air conditions such as ozone and salt
- Thermal cycling (especially relevant for assemblies, joints and complex parts with varying thicknesses, overmolded sections and intricate ribbing patterns)
- Short- and long-term properties at varying outdoor temperatures (Typical outdoor design criteria consider temperatures of -40-85°C for most industrial applications; critical applications may require temperatures as high as 125°C and as low as -55°C.)
- Chemical compatibility (Chemical exposure in outdoor environments can include pesticides, acid rain and cleaners.)
With a thorough list of specification requirements, you can follow a systematic process of elimination that will result in a short list of suitable material alternatives. Your focus can then shift to optimizing photo-oxidation resistance. Numerous alternatives can be considered to provide your product with the necessary UV resistance. These may include:
- Opting for a polymer with inherent UV resistance.
- Adding a proper stabilization package to enhance UV resistance.
- Adding fillers such as carbon black or titanium dioxide that can enhance UV resistance.
- Protecting or coating the surface with a UV-resistant layer for outdoor applications (ABS can be chrome plated or painted; non-aliphatic polyurethanes are normally painted in outdoor applications (PC’s long-term weatherability is enhanced with siloxane hard coats.)
With such diverse alternatives, it’s best not to limit material considerations to only inherently UV-stable polymers. However, you must recognize that environmental stressors often act synergistically, accelerating degradation and increasing the likelihood of brittle failure and cracking. For this reason, it’s best to perform validation testing under realistic environmental conditions, ideally involving multiple stressors.
For instance, when a polyamide is exposed to high outdoor moisture, its properties will be significantly different than when the material is without significant moisture. Most designers understand the response of strength to moisture, but other long-term properties such as creep resistance will also be greatly affected (Figure 2). Similarly, aliphatic polyester-based thermoplastic polyurethane (TPU), though marketed as UV stable due to its aliphatic nature, may degrade under the presence of high humidity and heat (Figure 3). When performance data is lacking for a candidate material, the best course of action is either to select a better-characterized alternative or to validate the specification requirements through necessary testing.
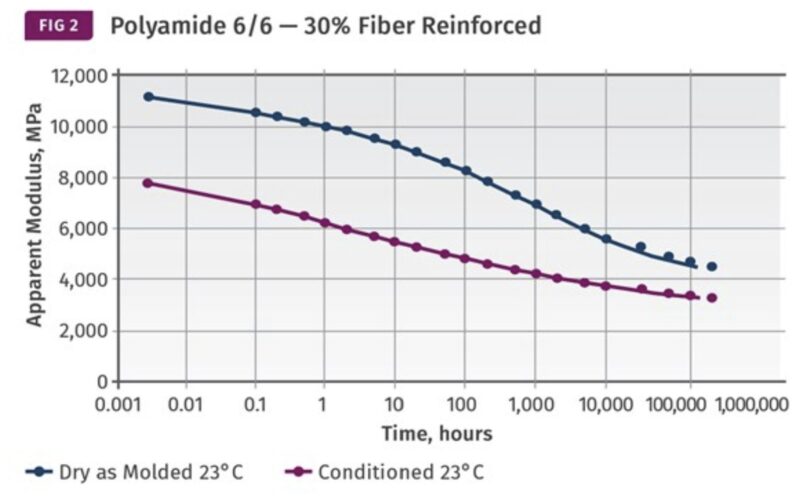
Figure 2: Long-term creep properties of polyamide 6/6 with and without moisture.

Figure 3: A fractured surface of a severely degraded polyester-based TPU was damaged from exposure to heat and humidity.
Fit for the Great Outdoors
While many polymers today are marketed as suitable for outdoor use, the optimal material choice will be the one that best meets the comprehensive specification requirements of the application. It’s crucial to fully establish these requirements early in the development process to maximize the likelihood of long-term success. All too often, the urgency to bring a product to market compromises the time available for proper material verification and validation, leading to unforeseen failures. A deliberate approach, driven by addressing all critical requirements, will ultimately yield a more reliable and robust product for outdoor environments.
This article was originally published in the October 2025 issue of Plastics Technology Magazine.
