The characteristic properties exhibited by plastics are the direct result of the unique molecular structure of these materials. Taking that a step further, the variation within the properties demonstrated by different plastics arises from diversity in their structure. Plastics are polymers of very high molecular mass. To enhance their properties, they often contain additives, such as fillers and reinforcements, anti-degradants and stabilizers, flame retardants and plasticizers. However, the underlying attributes of a plastic material are determined by the polymer.
The key molecular structure characteristics of plastics include: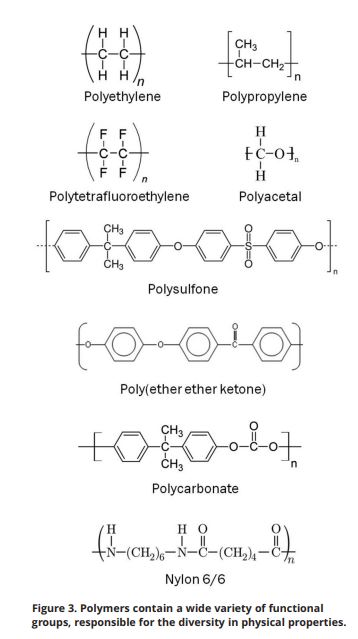
Polymerization
Polymers are macromolecules that are based on a structure built up, chiefly or completely, from a large number of similar structural units bonded together. Often called chains, the polymer consists of repeating units, similar to links. Polymers are formed through a process known as polymerization, in which monomer molecules are bonded together through a chemical reaction that results in a three-dimensional network of long individual polymer chains consisting of smaller repeated units.
Intermolecular Bonding
As indicated, polymerization results in the formation of multiple individual polymer chains made up of repeating units. A key aspect of polymeric materials is that the chains are entangled within each other. The individual chains are not covalently bonded to each other, but instead rely on intermolecular forces, such as Van der Waals forces, hydrogen bonding, and dipole interactions, to keep the chains from disentangling. This results in a structure that is similar to a bowl of spaghetti noodles.
Molecular Weight
Through the polymerization process, materials of relatively high molecular weight, macromolecules, are produced. A key parameter of a polymer is its molecular weight. Molecular weight is the sum of the atomic weights of the atoms comprising a molecule. For example, the molecular weight of polyethylene is calculated by multiplying the molecular weight of the repeating ethylene functional group times the number of units comprising the chain. Thus, for polyethylene, where the repeating unit contains two carbon atoms and four hydrogen atoms, the molecular weight is 28n, where n represents the number of repeating segments. Most commercial polymers have an average molecular weight between 10,000 and 500,000.
Crystalline/Amorphous Structure
Another fundamental characteristic of polymeric materials is the organization of their molecular structure. Broadly, plastics can be categorized as being semi-crystalline or amorphous. Understanding the implications of the structure, and specifically, the crystallinity, is important as it affects material selection, part design, processing and the ultimate anticipated service properties.
This article originally appeared in Plastics Engineering Magazine – September 2016
