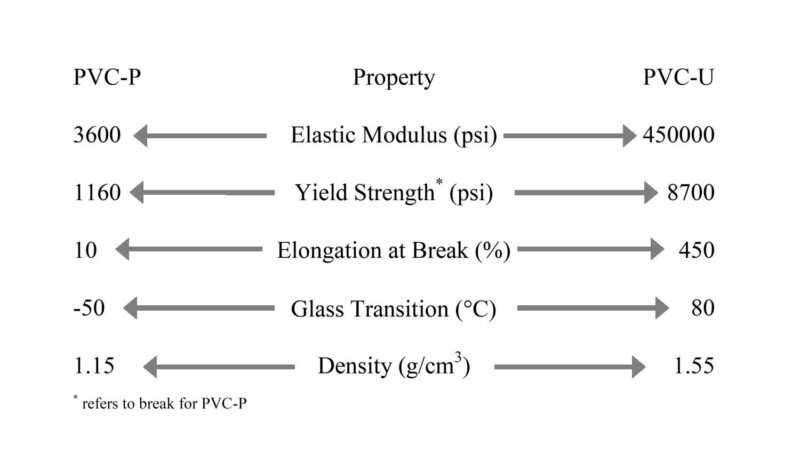
Figure 1 – Schematic showing the vast property ranges for PVC.
Poly(vinyl chloride) (PVC) stands out from most other plastics due to its truly unique characteristics. These qualities make PVC one of the most widely used plastics globally, with its PVC composition allowing for an incredibly diverse range of products manufactured through numerous processing techniques. It’s remarkable to consider that you might be standing on a PVC (vinyl) floor, looking through a window with PVC frames, while opening a faucet that drains water through PVC pipes. You could even be observing a neighbor’s house, which likely features PVC siding or a PVC fence. Most people don’t realize it, but PVC is almost ubiquitous. Beyond its abundance in construction materials like piping, siding, flooring, and wire, PVC can also be found in various other applications such as packaging, household products, toys, and medical devices. The beauty of PVC composition lies in its ability to be formulated into materials ranging from hard and tough to soft and flexible. As Figure 1 illustrates, the potential range for the mechanical properties of this material can be extremely broad, all depending on its specific formulation. So, what gives PVC the capacity for such a vast array of properties? To understand this, one must grasp the fundamental principles of PVC composition.
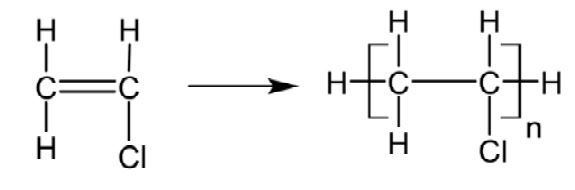
Figure 2 – Chemical reaction for PVC polymerization.
The Basic Structure of PVC Composition
PVC is manufactured from vinyl chloride monomer via a free radical reaction to produce a polymer with a simple backbone structure as shown in Figure 2. The structure looks much like a polyethylene except a chlorine atom replaces hydrogen resulting in a chlorine content of approximately 55% by mass. The chlorine atom is much larger than a hydrogen atom which will greatly affect the stiffness of the backbone. Additionally, a halogen atom on the backbone will also provide a unique set of chemical resistance properties, a crucial aspect of PVC composition.
Like all plastics, PVC properties can be modified by changes in its chemical structure. Molecular weight (MW) is a critical element directly related to the molecular structure that profoundly impacts mechanical properties and processing behavior. Molecular weight values are controlled at the material synthesis level, but they can also be affected during processing and while parts are in service. A reduction in molecular weight due to aggressive processing or chemical attack can lead to brittle material behavior. Other modifications that can be made directly to the PVC’s molecular structure include controlling the position of the chlorine atom or increasing the chlorine content. The latter results in another popular material, CPVC, which is a testament to how slight variations in PVC composition can create distinct polymers. However, this is a topic for another article.
 While specific changes in the polymer’s chemical structure can affect material properties, it’s generally not the preferred technique for controlling most properties of PVC. The use of additives is the primary and most effective way to obtain PVC material with the best processing and performance characteristics. In general, modifying the polymer structure itself only enhances certain properties, not all the necessary ones to produce reliable products. The clever use of additives, however, can enhance most properties of interest, typically at a fraction of the cost, making it central to successful PVC composition.
While specific changes in the polymer’s chemical structure can affect material properties, it’s generally not the preferred technique for controlling most properties of PVC. The use of additives is the primary and most effective way to obtain PVC material with the best processing and performance characteristics. In general, modifying the polymer structure itself only enhances certain properties, not all the necessary ones to produce reliable products. The clever use of additives, however, can enhance most properties of interest, typically at a fraction of the cost, making it central to successful PVC composition.
The often-overlooked truth is that PVC on its own is of limited practical use. Raw polymerized PVC is a material that is extremely difficult to process. It exhibits poor heat stability and a high coefficient of friction, causing the polymer to stick to the metal surfaces of processing equipment. If one were to process 100% PVC, the result would be overly brittle parts with very poor mechanical performance. So, what transforms PVC into such a versatile material? It is the intricate combination of numerous additives that form a PVC compound, making PVC composition a complex art.
The Art of PVC Composition: Additives as Key Ingredients
PVC composition is truly a “recipe” of numerous additives, such as fillers, stabilizers, lubricants, and processing aids, all compounded together to create a plastic with unique processing and performance characteristics. The ability to utilize numerous individual additives provides the immense benefit of tailoring the material for a specific processing technique and application. In this article, we will discuss several of the most commonly used additives for improving the mechanical performance of the final product. Additives specifically intended for affecting the processing of the material, as well as processing effects, will be discussed in the next article.
Common additives designed to improve multiple properties in the final product through their inclusion in PVC composition include plasticizers, UV stabilizers, impact modifiers, reinforcing agents, and flame retardants. As evident from its chemical structure (Figure 2), PVC is a highly polar polymer, which makes it compatible with multiple other high-performance polymers. Therefore, blending other polymers into the PVC mixture is a common technique for improving an otherwise poor material performance. Impact modifiers are a prime example of this strategy in PVC composition.
Impact modifiers, as expected, increase toughness and prevent brittle fracture response. Common examples of these modifiers include polymers such as ABS, CPE, EVA, and MBS. When mixing polymers like these, other properties can also be improved. For instance, ABS will also enhance heat resistance, while CPE, EVA, and MBS improve processability. Many other polymers can also be added to the blend to improve other properties. For example, TPU can be mixed into PVC to increase resistance to abrasion and material elasticity. All these polymers need to be properly compounded within the PVC to result in a homogeneous blend and optimize the material performance, highlighting the importance of precise PVC composition.
 Many fillers and reinforcing agents are currently used with PVC. These not only reduce cost but can also increase stiffness and enhance the shrinkage properties of the material. Common fillers in PVC composition include minerals such as calcium carbonate, kaolin, and talc. Reinforcing agents can range from natural fibers to more complex reinforcements such as glass microspheres. In general, these agents can significantly improve mechanical properties such as tensile strength, flexural modulus, and deflection temperatures.
Many fillers and reinforcing agents are currently used with PVC. These not only reduce cost but can also increase stiffness and enhance the shrinkage properties of the material. Common fillers in PVC composition include minerals such as calcium carbonate, kaolin, and talc. Reinforcing agents can range from natural fibers to more complex reinforcements such as glass microspheres. In general, these agents can significantly improve mechanical properties such as tensile strength, flexural modulus, and deflection temperatures.
Plasticizers represent a unique set of additives that unlock a whole new range of applications for PVC. We could write an entire thesis on the complexities of these additives and their interactions with the polymer within the context of PVC composition. While different theories explain the interaction between the plasticizer and the polymer, the end result is the same: plasticizers interact with the polymer, leading to a softening effect. Plasticizers are chemicals that exhibit good affinity with the polymer. When PVC is plasticized, the molecular weight should not be affected. Plasticizers induce chemical effects that do not cause chemical attack (i.e., chain scission), but their affinity with the chemical used as a plasticizer increases molecular mobility.
Plasticization can be undesirable if the chemical has too much affinity (too aggressive) or in certain applications where the plasticizing chemical is unintentionally absorbed by the polymer, leading to a significant reduction in mechanical properties. The key characteristic of a plasticizer is its ability to interact with the polymer-to-polymer molecular forces, reducing them to soften the material. However, it cannot be too aggressive, or it will otherwise solvate the polymer. Furthermore, the best plasticizers are those that can interact sufficiently with the polymer chains so that they are not readily and easily diffused (leached) out of a part while in service, ensuring the long-term integrity of the PVC composition.
Plasticizers are added to the polymer under specific conditions of heat and stress. Therefore, exposure to these conditions, as well as contact with unplasticized materials of similar affinity, can allow for the plasticizer to diffuse out of the part. Currently, polymeric-based plasticizers are commonly used when there is an interest in limiting plasticizer diffusion, as it is more difficult for larger molecules to move freely. For example, for medical devices, certain phthalate-free alternative plasticizers have been developed due to increasing concerns about the possibility of plasticizers leaching out of the plastic and into the human body, influencing the choice of PVC composition for such sensitive applications.
 For PVC, the most common types of plasticizers used worldwide are phthalates. The most common types of phthalates used include DEHP (also known as DOP), DINP, and DIDP. Stronger plasticizers that cause a solvation effect on PVC typically exhibit high polarity and/or aromatic rings. When a plasticizer is absorbed by the polymer, the interaction with the polymer that leads to softening effects can be observed as a reduction in the glass transition (Tg), modulus, and material density. This allows for an otherwise stiff and brittle material to behave in a flexible and moldable manner, opening up an entirely new set of applications for PVC material, all thanks to its adaptable PVC composition. Very common applications for flexible PVC include cable and wire, medical devices, tubing, and soft toys. One of the great benefits of most of these plasticizers is their whitish color, which does not significantly affect the material’s color, allowing for transparent components to be manufactured.
For PVC, the most common types of plasticizers used worldwide are phthalates. The most common types of phthalates used include DEHP (also known as DOP), DINP, and DIDP. Stronger plasticizers that cause a solvation effect on PVC typically exhibit high polarity and/or aromatic rings. When a plasticizer is absorbed by the polymer, the interaction with the polymer that leads to softening effects can be observed as a reduction in the glass transition (Tg), modulus, and material density. This allows for an otherwise stiff and brittle material to behave in a flexible and moldable manner, opening up an entirely new set of applications for PVC material, all thanks to its adaptable PVC composition. Very common applications for flexible PVC include cable and wire, medical devices, tubing, and soft toys. One of the great benefits of most of these plasticizers is their whitish color, which does not significantly affect the material’s color, allowing for transparent components to be manufactured.
Analysis techniques are available for examining PVC composition and molecular changes within the polymer. These techniques can provide valuable insight into the processing characteristics and final properties of the polymer.
As discussed in this article, the PVC composition of a formulation is absolutely critical to obtaining the best performance for a given application. In Part 2 of this series, we will dive into processing, which is another critical aspect that directly affects the performance of PVC.
See the other installments in this series on PVC:
